CRDS Water Isotope Analysis - δD δ18O - Abel
EXPIRED - USE the generalized CRDS procedure.
Introduction
Abel is a Picarro L2130i liquid water cavity ring-down spectroscopy (CRDS) laser. dD and d18O of water are derived from absorption of specific spectral lines in the near-infrared range. Water vapor is introduced into an optical measurement cavity with an effective path length of up to 20 kilometers. We have three other Picarro liquid water isotope analyzers, Gorky, a L1102i, and Dr DeSoto, a L2120i, that both measure dD and d18O and Phoenix, a L2140i, that measures dD, d18O, and d17O.
Currently, "High Throughput" mode injections take 4.5 minutes each while "High Precision" mode injections take 9.3 minutes each. A full tray of 54 vials with 10 injections each, then, will take 1.7 days and 3.5 days for High Throughput and High Precision, respectively. Note that this method was originally written for DeSoto and the video links feature DeSoto. Some of the content in the videos is not applicable for Abel.
Safety
Users must be careful with syringe needles and vials. If either of these items are no longer useful (i.e. broken), they must be discarded into the broken glass container. The vaporizer is 110 °C and the septum nut can be hot to touch.
Terse Procedure
- Clean up previous run – close the Coordinator software, remove existing vials.
- Load new waters - load 200 µL of water into 300 µL fused insert vials.
- Take notes - write the tray description and any other notes in the notebook while loading waters into vials.
- Prepare autosampler - put the vials in the tray, clean the syringe, modify the job, don’t change the septum yet.
- Fill out the daily log which is bookmarked on the Abel computer.
- Start Run – Start Autosampler, open Coordinator Launcher, select High Precision or High Throughput, click Change Septum, wait for first injection to complete.
- Take more notes – write down time and date of run start, what you did with the syringe, septum etc.
- Change Septum – when the Coordinator indicates the first injection is complete, press “Septum Changed” when done.
- File management – make sure a directory has been created on the server for these samples (e.g. S:\data\projects\...).
- Tray Description – create a tray description file in the newly created folder, use a text editor, save as yymmdd_TrayDescription.csv.
- Data - use Matlab and the gorky script to reduce data from Abel.
- Cleanup - empty the existing vials, throw the caps in trash, discard water down the sink, put vials in a beaker in the drying oven, clean vials if needed.
Exhaustive Protocol 
Clean up previous run 
Close the Coordinator...be patient, it will close eventually. Remove vials from the previous run and set them aside for later cleanup.

If the instrument was not previously running AND the water backgrounds are currently in the 10,000 to 20,000 range, the valve on the back of the vaporizer (see photo) is allowing room air to flow into the instrument. Rotate the valve 180° to allow dry air to flow into the instrument. You should see the water backgrounds begin to drop. Ideally, the water background is below 100 ppm before you start.
Load new waters 
Load 200 µL of water into 300 µL fused insert vials. Use the pipette, pipette tips, and trays in 303B drawers labeled ‘Laser’. Vials are located on the lab bench above these drawers. Should you wish to load 1 mL of water, old, 2 mL vials without the 300 µL inserts which are located in a drawer labeled ‘Laser – extras’.
Use one full tray of dry clean pipette tips and also have one empty pipette tray available. For each distinct water, use a clean dry pipette tip, discarding the wet tips into the tray that started out empty. In this way, you will have one pipette tip tray with dry clean tips and one tray to receive the wet tips. If the pipette itself becomes wet, either on the inside or outside, ensure it is dry before proceeding. Use wipes or paper towels and / or compressed air to dry it out.
Make sure the caps on the vials are just tight enough but not too tight. If the cap septum is overly concave, it is too tight. Put all standard waters and all sample waters back in the refrigerator when loading is complete. Put the wet pipette tip tray in the drying oven. As you are loading waters, write the water type or sample ID in the notebook.
Lastly, when the 300 µL inserts are used, a tiny bubble is left at the very bottom of the insert after pipetting your water. This bubble must be dislodged. Hold the cap tight in one hand and flick the bottom of the vial with your other hand to jostle the bubble loose. Assume this is to be donw with every single vial.
Take notes
Write the date, your name, sample set ID, and the complete tray description as well as any other notes you desire in the notebook (Abel) while loading waters into vials.
Prepare autosampler
Vials - Place the tray of samples and standards onto the tray-holder, or, if you used a blue plastic tray to load your samples, transfer the vials from the temporary blue tray to the metal tray on the autosampler. If you move the metal tray at all, make certain it is properly seated. Visually check the caps one last time; no cap should be overly concave or crooked.

Remove the syringe - Whether you are going to clean the syringe or replace with a new one, you will need to remove the existing syringe. You can either just remove the syringe where the autosampler resides (preferred) or press "Chg Syringe", on the autosampler control software. CAUTION - Often times pressing "Chg Syringe" seizes the plunger in the barrel of the syringe. Remove the syringe from the autosampler by adjusting both black toggles, one at the plunger and one at syringe-body. Use the photo to help you identify the toggles. The top plunger toggle twists while the bottom syringe-body toggle pulls out and twists.
Clean the syringe - Find the syringe cleaning kit. Fill the 'helper' syringe that is in the kit (not the one you just removed in the above step) with the 1% HCl solution and set aside. Place the syringe you just removed from the autosampler into the HCl solution and pull the plunger up half way or as far as you can. Use the 'helper' syringe and apply HCl to the top of the autosampler syringe (e.g. where the plunger meets the syringe glass body - see photo). Set the helper syringe aside. Continue moving the autosampler syringe up and down while it is in the HCl solution until plunger can be removed from syringe body (this usually takes a while and you must be patient and gentle). Wipe off plunger with a wipe. Reinsert the plunger into the body and continue moving the plunger up and down while still in the HCl. Remove the plunger again and wipe it off. Inject all HCl back into HCl container. Fill the autosampler syringe with DI water by pulling plunger all the way to the top. Release this water into a wipe, and repeat ~5 times. Rinse heavily by moving the plunger up and down with the same DI water while keeping syringe tip in the DI water. Reinsert the, now clean, syringe into the autosampler and indicate COMPLETE on the autosampler controller. Rinse the 'helper' syringe with DI water in the same way. If you can’t remove the plunger during cleaning, break it, or it was already broken, obtain a new 10 µL syringe from the drawer labeled "Autosampler Syringes" to the right of the fume hood in 302A and near the IsoLab office. Discard the old syringe to the broken glass box in 303B under the northernmost sink (since this is not a skin piercing style needle, the broken glass box is sufficient)


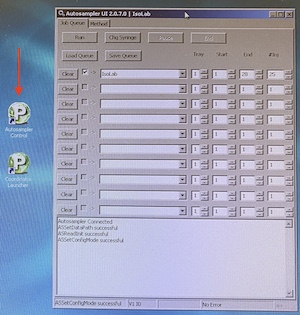
Autosampler job - Modify the autosampler job according to your current number of vials and your preferred number of injections per vial. If the autosampler control software is not open already, use the shortcut on the desktop to open it. Select "IsoLab" in the method field unless you have reason to pick a different type. The method is the way each individual injection is treated. Tray should always be "1". Start will typically be "1". End will be the position of the last vial. The #Inj is the number of injections per vial.
Daily Log 
The lab uses a daily log for each instrument or preparation line to allow users a first glance at the readiness of the instrument. By comparing the current state of the instrument to historical states, you are more informed about the instrument and whether or not it is functioning properly and ready to run your samples.
Each daily log is web based and browser accessible. No link is provided here by design. Open the browser on the controlling computer and you should see at least two tabs already open. One tab is this SOP and the other is the daily log. If the browser has more than two tabs open, it may have additional SOPs. Use the bookmark toolbar as needed if tabs have been closed.
Work through each cell of the daily log. If you are uncertain where to find certain information, hover over the column header tip, denoted by a question mark ( ⓘ ).
High Precision vs High Throughput - As part of the Daily Log, you will be asked to indicate if the runs is High Throughput or High Precision. These two modes of analyzing samples are aptly named. High Throughput mode aims to analyze many samples quickly and each injection takes 4.5 minutes. High Precision mode aims to obtain better precision and injections take 9.3 minutes each. A full tray of 54 vials with 10 injections each, then, will take 1.7 days and 3.5 days for High Throughput and High Precision, respectively. The difference between these two methods is the duration of time the instrument spends measuring and cleaning out each injection.
You will also be asked to indicate if you have replaced the vaporizer septum with a new one. Unless you have good reason not to, you will replace the old septum with a new one after you start the run below.
Make certain to press the 'save to log' button when you are finished entering data.
You are welcome to make notes if you have observed something with or done something to the instrument and would like to document that information. Use the "insert note" link at the top of the daily log to make a note. You may enter notes at any time.
Start Run 
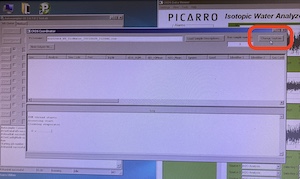
Click "Run" on the Autosampler Control software and wait for the autosampler to proceed through a series of initiation steps. Next, open the Coordinator Launcher from the shortcut on the desktop and select High Precision or High Throughput. Click "Change Septum" in the upper right corner of the Coordinator window and wait for first injection to complete.
Tray Description 
A good time to create the tray description file is while you are waiting for Abel to be ready to have its septum changed. The tray description file is used by the Coordinator software to assign each injection an Identifier which you give to each vial. As a template, use C:\Abel\TrayDescriptions\_TrayDescription.csv. Use a text editor to open one of these templates. If you double click on one of these templates, it will open in Notepad++. The header row in these templates is: "Tray","Vial","Identifier 1","Injections". The Tray and Vial columns are already filled in. Type in the water that is in each vial under Identifier 1. Example Identifier 1 entries are "SW", "WW", or "VW", without quotes. Type the number of injections under Injections. This may seem redundant, and hopefully it is, but sometimes when the autosampler fails or an injection is otherwise compromised, adjusting the number of injections in this file after the run has completed will help in data reduction. Save this file as yymmdd_[SampleSetID]_TrayDescription.csv (where yymmdd is today's date, and [SampleSetID] is optional and the square brackets are not included). The "TrayDescription" part of the filename is essential for some parts of the matlab code. Import your tray description file by clicking ‘Load Sample Description’ in the Coordinator software and then browse to the tray description you just saved (c:\Abel\TrayDescriptions\).
Change Vaporizer Septum 


After the first injection is complete, the coordinator will indicate when it's appropriate to change the septum. Remove the magnetic cover and use the slotted wrench side of this cover to loosen the nut (see photo). Remove the nut with your fingers, taking care as it is hot. The septum should be stuck inside the nut. Use the large-gauge needle that is in the syringe cleaning kit to extract the septum (see photo). Discard the old septum into the trash. Place the new septum into Swagelok nut using the blunt end of large-gauge needle. Place the nut back onto the vaporizer, taking care not to allow the septum to fall out when you turn the nut over. Hand tighten the nut; you should feel the nut bottom out. Press “Septum Changed” which is the same button you pushed to begin changing septum in Coordinator window.
Take more notes
Write down the time and date of run start. This is essential as files are named within the instrument based on time. Also write down what you did with the syringe, the septum, etc.
Data 
The Picarro instruments provide data at at least four different levels describe in Schauer et al (2016): "(1) coordinator data, (2) user data, (3) private data, and (4) spectral data. The coordinator data are what most users post-process to VSMOW-SLAP scales and publish; the coordinator file contains a single row of data for every injection and is readily imported into a Laboratory Information Management System (LIMS) such as LIMS for Lasers 2015. Each datum in the coordinator data is the average, variance estimate, or otherwise-summarized value across the 120–500 s duration of usable data from an injection, and originates from the user data. The user data file is typically used if the pulse analysis fails and a user employs the pulse analysis software to redefine the peaks. The private data are high-resolution unprocessed data with all instrument parameters recorded at a frequency of approximately 1 Hz but are still a reduction from the spectral data. The spectral data are the measured absorbances during each ring-down and have a frequency of approximately 500 Hz".
There are several ways you can proceed with your data analysis: DIY, easy, or comprehensive.
- DIY - Of course you are welcome to take the raw data at any of the above mentioned levels and run with it.
- Easy - This approach is easiest because it takes a single small file (produced by the Coordinator software) with only the most salient data and, using a Matlab script, provides you with water isotope values normalized to the VSMOW-SLAP scale. Coordinator data files are written to C:\IsotopeData\ in comma delimited value (.csv) format. Copy this file to your preferred project folder on the data server or to S:\Data\projects\abel\raw\. Move to the North, South, or West Going ZaxThe naming of these three computers was inspired by Dr. Seuss' story about the North Going Zax and the South Going Zax (shown in the image). There is no West Going Zax in his story, but the same idea applies.
 computer. Open Matlab, type abel and follow the prompts. After the script completes, the exported file is in S:\Data\projects\abel\reduced\ or the reduced folder of your preferred project directory.
computer. Open Matlab, type abel and follow the prompts. After the script completes, the exported file is in S:\Data\projects\abel\reduced\ or the reduced folder of your preferred project directory. - Comprehensive:
- abelh5.m - The abelh5.m script (1) defines the run, (2) finds all the injections, (3) creates injection level data by summarizing (mean, standard deviation, etc.) the high resolution data within each injection or pulse, and (4) creates vial level data by summarizing all injection level data for all injections in each vial. While you may wish to go back and do more diagnostic analyses with the high resolution data or the injection level data, it is the vial level data that most will be interested in. Make sure you have all files copied from the abel computer to the data server. After starting the script and selecting the main run folder (e.g. 150114_BradSnowpitA2), all files will be unzipped and then all h5 files will be loaded (this takes several minutes). Matlab then generates a figure to determine the start and end point of the run using water concentration data. Zoom in on the graph and arbitrarily select points about an injection width left of the first injection and right of the last injection. Enter start and end point at the prompt (e.g. 10900,649000). The script will then finish on its own. Created files are *_StartStop.mat, *_h5.mat, *_inj.mat, *_vial.mat, where * is the date in yymmdd format. You can plot the injection level H2O concentration data, for example, by plot(inj.h2om, ‘.’ ), where m is mean. H2O concentration should be very near 20,000 ppm. You can also look over d18O or dD by plot(inj.d18Om, ‘.’ ). If you see points at the end of the run that are exactly zero, not all of the peaks have been identified or some error has occurred with the anticipated number of peaks.
- abeldrift.m - The abeldrift.m script is used to check for drift in vial level data over the course of the run. This script uses the _vial.mat file that was created using abelh5.m. Run abeldrift and select the *_vial.mat file that was created in the folder you are currently working in. You will need to tell the script what standards you want to use to drift correct. These are very likely the same standards you will use to calibrate. A file *_vial_drift_corr.mat was created into the S:\Data\projects\abel\driftcorrected\ folder.
- abelcalibrate.m - The abelcalibrate.m script calibrates all vial level data to the standards included in your run. This can be done on a single run (drift corrected or not) or can be done with many runs. Make sure you have all the *_vial_drift_corr.mat files you want in a single folder before you start this script. A calibrated data file is created and placed into S:\Data\projects\abel\calibrated\.
- abelcompile.m - Finally, all calibrated data for a project can be loaded for plotting, summarizing, etc. using abelcompile.m. This script will load all calibrated .mat files from the folder you select and then create an exported summary file in S:\Data\projects\abel\calibratedexports.

Cleanup
Running more samples?
If you are not going to run more samples and you do not know of anyone after you wanting to run samples, rotate the valve on the back of the vaporizer (see photo above) 180° to allow room air to flow through the instrument rather than dry air. You should see the water backgrounds begin to increase.
Vials
Take vials with previously analyzed water to the wet lab. Open each vial, discard the caps into the trash, and discard the water down the sink by gently shaking the vials. If the vials contained dirty or salty samples, they must be soaked in DI water. You will have to pipette DI water into each vial. You may also use the vial-cleaning-hose-pipette-contraption as shown in image. Once vials are clean, gently place vials in beaker and put them into the drying oven. If soaking in DI water does not get the vial inserts clean, use the glass pipette rubber hose tool attached to a DI water spout to force DI water into the bottom of the insert.
Vaporizer Cleaning
This procedure is completed irregularly depending on the memory or carry-over from sample to sample. More salt buildup inside the vaporizer will leak to more carry-over from previous samples. Picarro wrote up a procedure for this. A pdf of this procedure is located under Resources in Manuals. We have slightly different parts to assemble the apparatus but the general workflow is the same. Also, consider using 5-10% acetic acid as the first rinse of the vaporizer. Follow this by numerous rinses with 18 MΩ water.
Troubleshooting
- The syringe has died - You come in to find the syringe mangled while the autosampler and instrument continue to run. Scroll up in the upper Coordinator window to find where the injections stopped working and note that vial number. Close the Coordinator. Replace the syringe with a new one. Discard dead syringe into the broken glass box located in room 303B under the northernmost sink (since this is not a skin piercing style needle, the broken glass box is sufficient). Modify the autosampler job to start on the vial you noted above. Start the instrument as normal, but don't worry about changing the septum.
- The water background is way too high - Check the valve on the back of the vaporizer to ensure it is pointing towards dry air (see photo at top of this page).
- ALS-G Autosampler Maintenance - [need to write more here, for now, link to picarro docs]. Lubricant doc, Z-axis spring doc, Lubricant maintenance doc
Suggested Reading
- Lis G, Wassenaar LI, Hendry MJ. (2008) High-precision laser spectroscopy D/H and 18O/16O measurements of microliter natural water samples. Analytical Chemistry 80, 287 doi: 10.1021/ac701716q.
- Guidotti S, Jansen HG, Aerts-Bijma AT, Verstappen-Dumoulin BMAA, van Dijk G, Meijer HAJ. (2013) Doubly Labelled Water analysis: Preparation, memory correction, calibration and quality assurance for δ2H and δ18O measurements over four orders of magnitudes. Rapid Communications in Mass Spectrometry 27, 1055. doi: 10.1002/rcm.6540.
- Gupta P, Noone D, Galewsky J, Sweeney C, Vaughn BH. (2009) Demonstration of high-precision continuous measurements of water vapor isotopologues in laboratory and remote field deployments using wavelength-scanned cavity ring-down spectroscopy (WS-CRDS) technology. Rapid Communications in Mass Spectrometry 23, 2534. doi: 10.1002/rcm.4100.
- Schauer AJ, Schoenemann SW, Steig EJ. (2016). Routine high-precision analysis of triple water-isotope ratios using cavity ring-down spectroscopy. Rapid Communications in Mass Spectrometry 30, 2059–2069. doi: 10.1002/rcm.7682.
- Wassenaar LI, Kumar B, Douence C, Belachew DL, Aggarwal PK. (2016) Measurement of extremely 2H-enriched water samples by laser spectrometry: application to batch electrolytic concentration of environmental tritium samples. Rapid Communications in Mass Spectrometry 30, 415. doi: 10.1002/rcm.7459.
- West AG, Goldsmith GR, Brooks PD, Dawson TE. (2010) Discrepancies between isotope ratio infrared spectroscopy and isotope ratio mass spectrometry for the stable isotope analysis of plant and soil waters. Rapid Communications in Mass Spectrometry 24, 1948. doi: 10.1002/rcm.5126.
Other Resources
Picarro has a series of tutorial videos that may be helpful.
